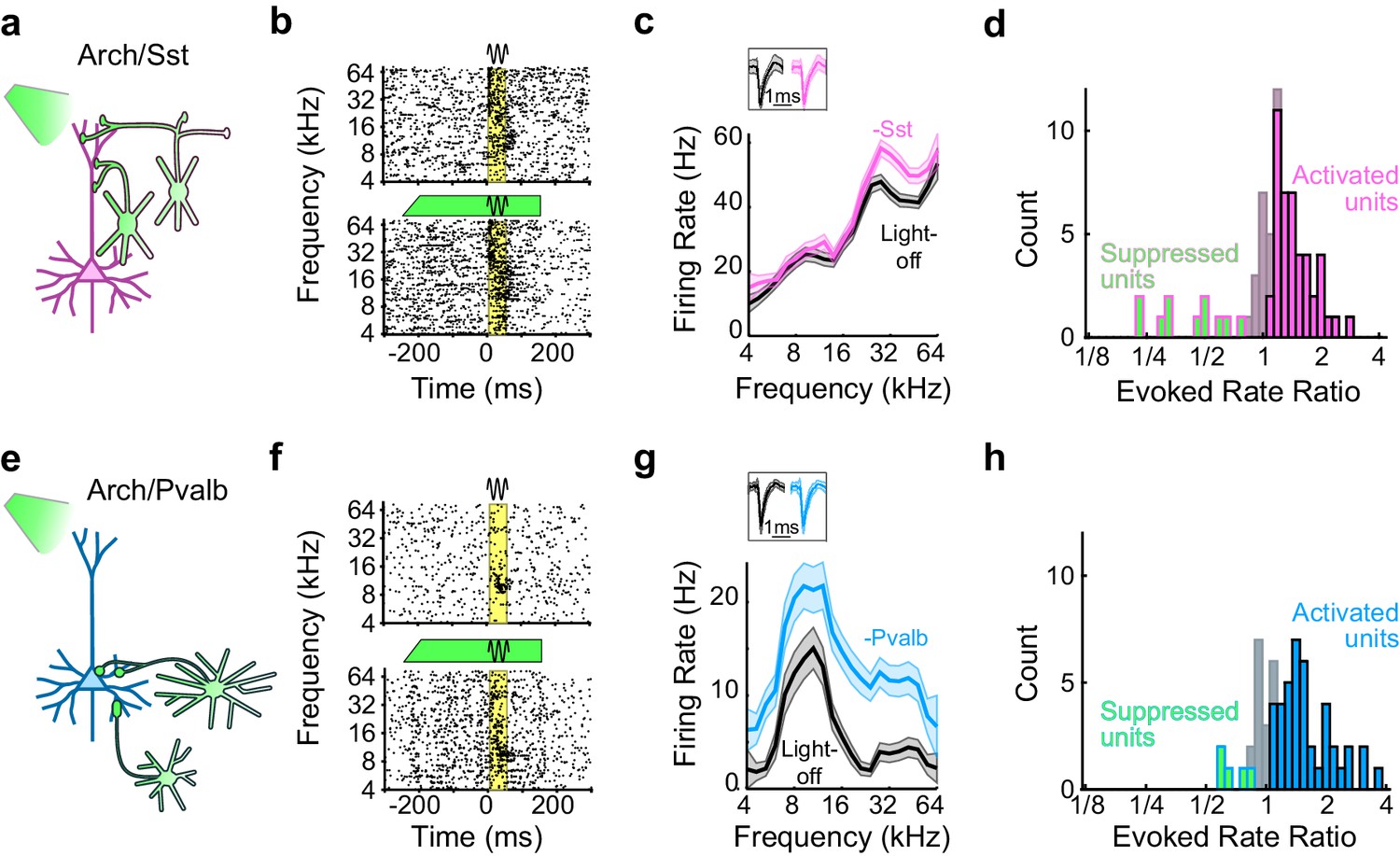
Phillips, EAK and Hasenstaub, AR (2016) Asymmetric effects of activating and inactivating cortical interneurons eLife 2016;5:e18383.
The Hasenstaub Lab at UCSF operates under Dr. Andrea Hasenstaub within the Department of Otolaryngology - Head and Neck Surgery and in the Center for Integrative Neurosciences. The lab focuses on understanding the genetic, cellular, and network operation of specific cell types in the mouse and human auditory cortex.
For any inquiries, please reach out to [email protected] or call our lab manager at (415) 502-7557 (or by email, [email protected]).
